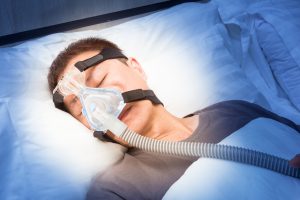
Philips Respironics has recalled multiple CPAP machines, PAP machines and Ventilators due to the risk of “serious injury which can be life-threatening, cause impairment, and require medical intervention to prevent permanent damage.” The defect stems from defective insulation that breaks down causing toxic fumes and particles to enter the respiratory tract and lungs of individuals most at need for respiratory support.
Philips has used this insulation in its respiratory support machines for well over a decade and issued a formal recall on June 14th, 2021. The official recall can be found on the FDA website under “Recalls, Market Withdrawals, and Safety Alerts.”
Which Philips Machines Are Being Recalled?
Not all Philips’ ventilators, CPAP, and PAP sleep apnea machines have been recalled. About 80% of the affected machines are CPAP machines and 20% are ventilators, most coming from the Philips Dreamstation line. The recall affects the following machines manufactured between 2009 and April 26th, 2021:
Recalled CPAP and BiPAP Machines:
- C-Series ASV
- C-Series S/T & AVAPS
- Dorma 400
- Dorma 500
- DreamStation
- DreamStation Go
- DreamStation ST, AVAPS
- E30 (Emergency Use Authorization)
- Noncontinuous Ventilator
- OmniLab Advanced+
- REMstar SE Auto
- SystemOne ASV4
- SystemOne (Q-Series)
Recalled Ventilators:
- A-Series BiPAP A30
- A-Series BiPAP V30 Auto
- A-Series BiPAP Hybrid A30 (not marketed in the US)
- A-Series BiPAP A40
- Garbin Plus, Aeris, LifeVent
- Trilogy 100
- Trilogy 200
What Makes Philips’ PE-PUR Foam Potentially Harmful?
When PE-PUR foam breaks down, it can release chemical gasses and particles into the air. These particles and gasses can enter the body through the nose or mouth and can cause serious health issues.
According to the US Federal Food and Drug Administration, “these health issues can result in serious injury which can be life-threatening, cause impairment, and require medical intervention to prevent permanent damage.”
The recall was issued after Philips received limited reports of health problems potentially related to foam inhalation/ingestion. Some reports included complaints of black particles of deteriorated foam contaminating the machines’ airpath circuits.
Potential symptoms from foam particle and gas inhalation or ingestion can include:
- Airway irritation and inflammation
- Headache
- Skin and eye irritation
- Asthma
- Cough
- Sinus infection
- Upper airway irritation
- Organ damage (especially kidney and liver)
- Carcinogenic (cancer causing) effects
Philips analysis of the deteriorated foam confirmed the presence of several potentially harmful chemicals including toluene diamine, toluene diisocyanate, and diethylene glycol. During the report, Philips also identified toxic gasses, dimethyl diazene and phenol, 2,6-bis (1,1-dimethylethyl)-4-(1-methylpropyl)- that exceeded the safe exposure threshold. The health risks associated with these gases, include:
- Airway inflammation and irritation
- Headache
- Dizziness
- Nausea
- Vomiting
- Skin, eye, nose, and respiratory tract inflammation
- Hypersensitivity
- Carcinogenic and toxic effects including cancer
Who Qualifies to File a Philips CPAP Lawsuit?
If you or a loved one has been injured, experienced health problems, or has passed away after using one of the recalled devices, you may be eligible to seek compensation through filing a Philips CPAP lawsuit in New York State.
Cancer From Use of Philips Devices
Philips has confirmed that one of the causes for the recall is the potential that exposure to the PE-PUR chemical exposure may cause or contribute to the development of cancer. Cancer types that are accepted for this investigation are:
- Bladder Cancer
- Brain Cancer
- Breast Cancer
- Hematopoietic (Blood) Cancer
- Kidney Cancer
- Liver Cancer
- Leukemia
- Lung Cancer
- Lymphatic Cancer
- Multiple Myeloma
- Nasal Cancer
- Non-Hodgkin Lymphoma
- Papillary Carcinoma
- Prostate Cancer
- Rectal Cancer
- Stomach Cancer
- Testicular Cancer
- Thyroid Cancer
Serious Injuries From Use of Philips Devices
Philips also confirmed the potential for many other health risks from PE-PUR chemical exposure including serious respiratory issues. If you have suffered from any of the following after using a defective Philips CPAP, BiPAP, or ventilator, you may be entitled to compensation:
- Acute Respiratory Distress Syndrome (ARDS)
- Chemical Poisoning
- Heart Attack
- Heart Failure
- Kidney/Renal Damage
- Kidney/Renal Disease
- Liver Damage
- Liver Disease
- Lung Damage
- Lung Disease
- Pleural Effusion
- Reactive Airway Disease (RAD)
- Respiratory Failure
- Severe Ear Inflammation
- Severe Nose Inflammation
- Severe Throat Inflammation
How a Personal Injury Attorney Can Help With Your Philips CPAP Lawsuit
Developing a serious injury or cancer can significantly impact your life financially, emotionally, and physically. The help of an experienced personal injury attorney is the best way to have your potential claim properly investigated and pursued. Compensation can include:
- Medical expenses
- Loss of wages
- Loss of future earnings
- Pain and suffering
- Wrongful death
If you have experienced health issues or have lost a loved one due to one of the defective Philips machines, there are time limits to file a claim. It is important to reach out and speak with an experienced attorney as early as possible to protect your rights.
At Cellino Law, we understand that filing a lawsuit against an international medical device company such a Philips can seem overwhelming and complicated. With over 60 years of experience in dealing with product liability and medical device defect cases, we are confident that we can assist you to achieve the best outcome.
Our attorneys at Cellino Law have successfully litigated against all major medical product manufacturers for decades. Our proven track record includes hundreds of millions of dollars in recoveries for clients injured by defective medical products and medications.
To begin your Philips CPAP lawsuit, reach out to Cellino Law 24/7 for your free case evaluation. With Cellino Law, you do not pay until we win for you. Call us today at 888-888-8888.